Overview
Antibody–drug conjugates (ADCs) require multi-analyte quantification to fully characterize pharmacokinetics and drug stability. This study implemented an integrated bioanalytical strategy to quantify total antibody (TAB), intact ADC, and free payload in biological matrices. The workflow combined electrochemiluminescence (ECL) ligand-binding assays and LC–MS/MS to achieve sensitive, selective detection across broad concentration ranges.
Challenges
- Multiple analytes requiring different analytical technique
- Need to differentiate intact ADC from unconjugated antibody
- Detection of free payload at very low (pg/mL) concentrations
- Wide concentration ranges and tight project timelines
Approach
- Total Antibody (TAB): Quantified using an antigen-capture ECL ligand-binding assay on the MSD platform with a dynamic range of 11–1600 ng/mL.
- Intact ADC: Measured using an ECL immunoassay with anti-payload capture antibodies, enabling selective detection of conjugated antibody species (4–210 µg/mL).
- Free Payload: Quantified using LC–MS/MS following optimized solid-phase extraction (SPE) to improve recovery and sensitivity (50–5000 pg/mL).
- Method development and sample analysis were conducted in parallel to accelerate project timelines.
Outcomes
Pharmacokinetics were assessed in male (Figure part A) and female (Figure part B) subjects across four doses (3, 10, 30, 60 mg/kg):
1TAB
Dose-dependent peaks up to 2.0 × 106 ng/mL (male; Figure 1A), slightly lower in females (Figure 1B), followed by multi-phase clearance over 21 days.
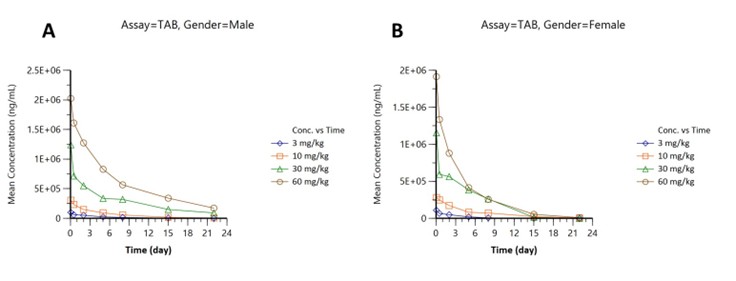 Figure 1: Flow cytometry analysis of CHO-K1-eGFP monoclones.
Figure 1: Flow cytometry analysis of CHO-K1-eGFP monoclones.
2Intact ADC
Similar dose trends with peaks 1.8 × 106 ng/mL (male; Figure 2A) and 1.3 × 106 ng/mL (female; Figure 2B), consistent elimination kinetics.
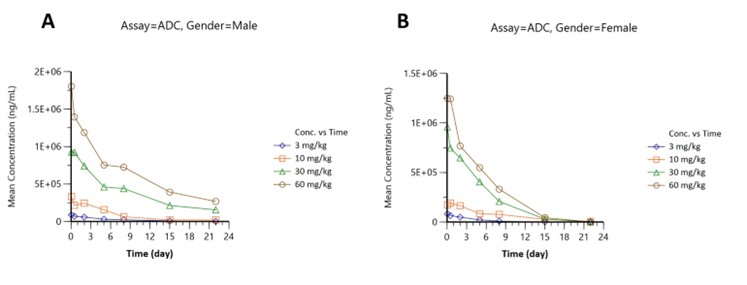 Figure 2: Total antibody (TAB)concentration-time profiles: (A) males, (B) females, doses 3–60 mg/kg.
Figure 2: Total antibody (TAB)concentration-time profiles: (A) males, (B) females, doses 3–60 mg/kg.
3Free Payload (Exatecan)
Sensitive detection at low ng/mL levels, peaks 2.3 (male; Figure 3A) and 2.1 ng/mL (female; Figure 3B), rapid clearance.
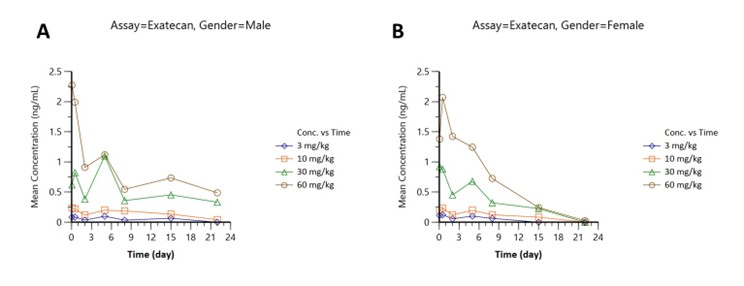 Figure 3: Free payload (Exatecan) concentration-time profiles: (A) males, (B) females, doses 3–60 mg/kg.
Figure 3: Free payload (Exatecan) concentration-time profiles: (A) males, (B) females, doses 3–60 mg/kg.
4Operational efficiency
Method development and bioanalytical sample analysis were completed within an accelerated timeline.
These results demonstrate the robustness and sensitivity of the integrated bioanalytical workflow, enabling comprehensive PK characterization across doses and genders.
Conclusion
This case study demonstrates that our integrated ECL and LC–MS/MS workflow delivers fast, sensitive, and reliable quantification of TAB, ADC, and free payload, enabling clients to accelerate ADC development and generate robust pharmacokinetic data.